Govern aRMM and Risk Commitments with Structured Intelligence
AI-Powered RMP and aRMM Management Software for Global Pharmacovigilance Teams
Quartica MARS streamlines RMP tracking to accelerate global and local commitment execution. It transforms aRMMs from manual trackers into governed, traceable outcomes – linking RMPs, implementation plans, and local assessments to materials, distribution, effectiveness tracking, and regulatory reporting.
EU Regulation 2025/1466: The Compliance Threshold Has Changed
Effective February 12, 2026, EU Regulation 2025/1466 requires that aggregate reports including PBRERs document not only the existence of risk minimization measures but measurable evidence of their effectiveness and practical implementation. For organizations still managing aRMM commitments through static trackers and disconnected documents, this is a direct inspection liability not a future consideration. Quartica MARS is built to meet this requirement by connecting commitments, materials, distribution, and effectiveness evidence within a single governed framework.
Beyond a Tracker: End-to-End RMP and aRMM Commitment Governance
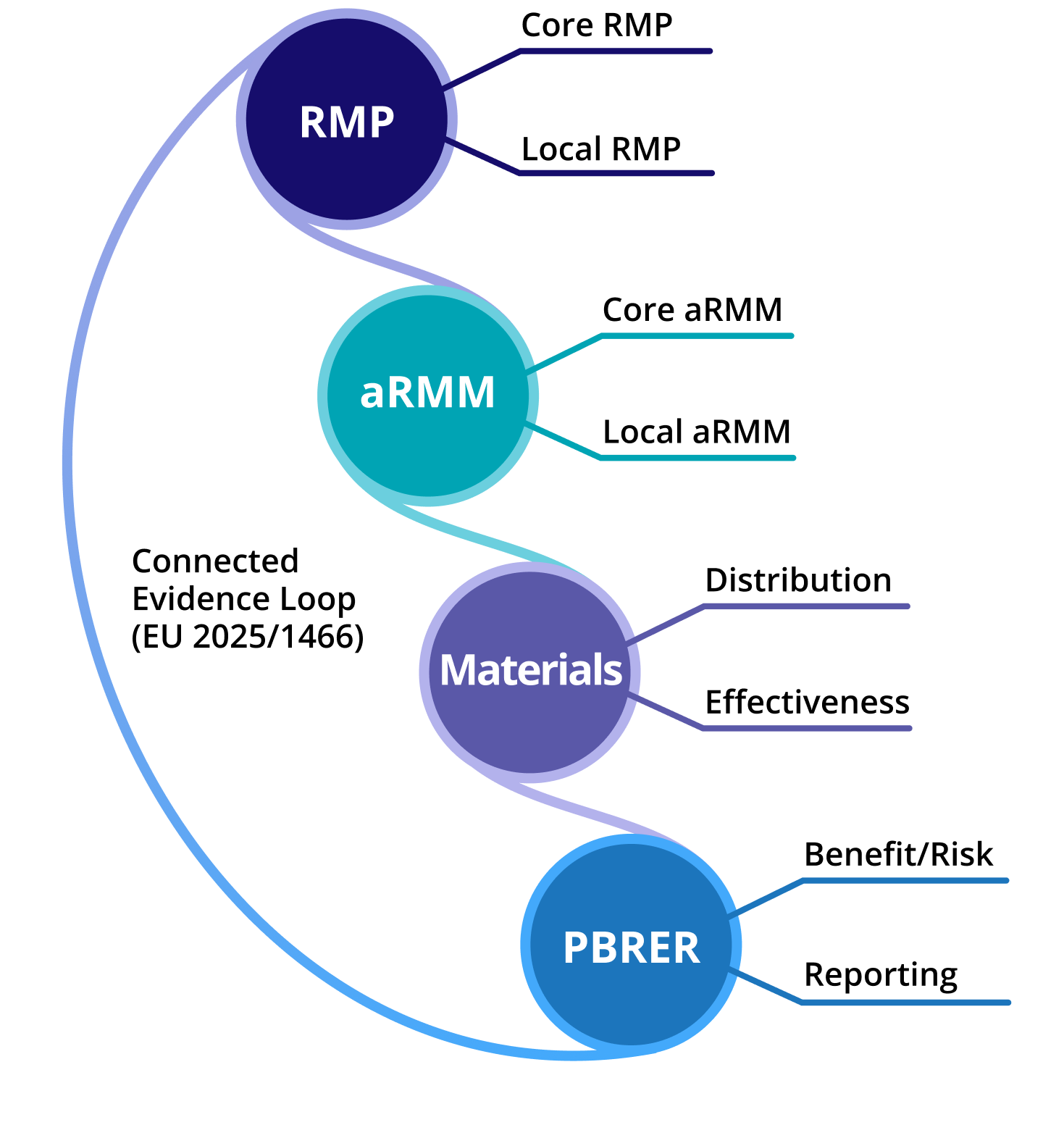
Risk minimization measures originate in Core and EU RMPs. Critically, they must cascade into Local RMPs, require the development of materials, and call for full traceability from controlled implementation through distribution. Ultimately, companies must be able to demonstrate measurable effectiveness.
Just a single RMP change, regardless of source, can impact multiple markets, commitments, materials, and reporting obligations leading to complex impact assessments.
With evolving regulatory expectations, including EU 2025/1466 requirements for structured aRMM effectiveness reporting within aggregate reports such as the PBRER, organizations must move beyond static tracking toward evidence-linked governance.
Yet many companies still manage these processes through a patchwork of:
- Static trackers and spreadsheets
- Disconnected RMP documents
- Separate material repositories
- Manual local updates
- Standalone effectiveness logs
- Manual impact analysis and QC reconciliation
The result is fragmented traceability, weak linkage between commitments and evidence, version confusion across Core and Local plans, and increasing inspection exposure. All of these represent risks that only intensify as pipelines and global complexity grow.
How Quartica MARS Manages RMP-to-aRMM Commitment Traceability
Quartica MARS is purpose-built as a validated global operating platform for risk commitments and not just as a tracker or workflow layer.
It embeds aRMM, aPV, DHPC and other pharmacovigilance activities within a relational governance framework that connects:
- RMP risk definitions
- Core and Local commitments
- Risk minimization materials
- Core and Local Implementation plans
- End-to-end distribution activities
- Effectiveness metrics
- Version-controlled change history
Complex relationships including Parent–child structures across EU RMP, Core RMP, Local RMP, Core aRMM, and Local aRMM are modeled natively. When an RMP changes, downstream commitments and materials can be assessed and governed within a structured framework, removing the need for manual reconciliation.
With its AI-driven authoring capabilities, MARS can generate full drafts of RMP, aRMM materials and automate the extraction of key information directly from RMP documents (risks, aRMMs, PV activities, etc.). The critical linkage between regulatory intent and operational execution is as a result preserved from the outset in real time.
The result is not simply tracking. It is controlled, inspection-ready risk governance aligned to modern regulatory expectations where commitments remain traceable, evidence remains linked, and effectiveness of reporting becomes a structured output rather than a manual exercise.
Did you know Quartica MARS offers solutions for PBRER, RMP authoring and PSMF?
Key Capabilities: RMP Commitment Tracking and aRMM Lifecycle Management
Structured Core-to-Local Governance
All RMP relationships natively preserving traceability across commitments, flow of information between core and local components, linking CIPs, and LIPs.
End-to-End aRMM and aPV Lifecycle Management
Govern commitment creation, versioning, material linkage, approvals, implementation oversight, updates, and retirement within validated workflows.
AI-Assisted Commitment Extraction & Authoring
Extract risks, aPV and aRMM elements directly from RMP content, and support structured RMP and material authoring , preserving linkage from regulations to operations.
Orchestrated Workflows and Task Management
Quartica MARS orchestrates tasks across global and local teams, automating reminders and providing consolidated views of all commitments for stakeholders.
Regulatory-Driven Change Control
When RMP updates or HA requests occur, assess downstream impact across markets, materials, and commitments before changes are executed.
Evidence-Linked Effectiveness Reporting
Connect KPIs, surveys, distribution, and monitoring to each commitment, enabling effectiveness reporting aligned with EU 2025/1466 and PBRER.
Enterprise System Integration
Secure, API-driven bidirectional integration with RIM, safety, quality, systems enabling structured data exchange.
Built-In QC & QPPV Oversight
Deliver dashboards reflect completeness of RMP, CIP, LIP, materials, and distributions activities supporting structured quality review.
Audit-Ready Reporting in Seconds
Respond to inspections with confidence. Every distribution record by country, version, and date is instantly accessible, complete, and fully traceable without manual effort.
Your next inspection will ask about aRMM effectiveness. Will your answer be immediate?
Frequently Asked
Questions
Is Quartica MARS just a commitment tracker?
No. Quartica MARS models structured relationships between RMP versions, Core and Local commitments, materials, implementation activities, and effectiveness evidence functioning as a validated system of record for global aRMM and aPV governance, fully synchronized with RMP documents. Unlike a tracker, MARS governs what actually happened across the full lifecycle, not just what was planned.
How does Quartica MARS manage Core-to-Local RMP and aRMM relationships across markets?
Parent-child structures across EU RMP, Core RMP, Local RMP, Core aRMM, and Local aRMM are modeled natively within the platform. When a Core commitment or material changes, downstream Local plans can be assessed and updated within a structured workflow preserving consistency between global strategy and local execution without manual cross-referencing or version reconciliation.
Does Quartica MARS support aRMM effectiveness reporting requirements under EU Regulation 2025/1466?
Yes. EU Regulation 2025/1466, fully applicable from February 12, 2026, requires that aggregate reports including PBRERs document not only the existence of risk minimization measures but measurable evidence of their effectiveness and practical implementation. Quartica MARS connects KPIs, distribution activities, stakeholder engagement, and monitoring outcomes directly to each commitment enabling structured effectiveness reporting as a governed output rather than a manual exercise, fully aligned with evolving EMA expectations.
How does Quartica MARS handle RMP updates triggered by Health Authorities?
The platform maintains version-controlled lineage between prior and updated RMPs, automatically identifies impacted commitments, materials, and Local Implementation Plans, and governs downstream updates within structured workflows. When an HA-triggered change occurs, teams can assess the full scope of impact across markets and commitments before changes are executed eliminating the manual reconciliation that typically follows an RMP revision.
What happens when a commitment or material is updated mid-lifecycle?
Quartica MARS preserves version-controlled lineage between prior and updated commitments and materials. Impacted Local Implementation Plans and distribution records can be assessed before changes are finalized reducing version confusion, preventing compliance gaps, and maintaining a complete audit trail of what changed, when, and why.
How does Quartica MARS support distribution tracking across Core and Local Implementation Plans?
Quartica MARS supports structured distribution models aligned to each Core and Local Implementation Plan, including direct dissemination, vendor-supported distribution, and manual Health Authority portal submissions. Distribution records are linked to commitments and materials for inspection-ready traceability so that every distribution activity is traceable to its governing commitment, version, and market without manual reconstruction.
Can Quartica MARS track HCP, investigator, and stakeholder acknowledgements?
Yes. HCP, investigator, or stakeholder acknowledgements can be tracked within the platform, including automated reminders, manual confirmation recording, and import of acknowledgement data from external systems where required by local regulations. Acknowledgement records are linked to the relevant commitment and distribution activity, supporting end-to-end traceability from dissemination through confirmed receipt.
Does Quartica MARS support follow-up and nested dissemination campaigns?
Yes. Quartica MARS supports nested distribution that preserves the relationship between initial and follow-up disseminations reflecting activity across distribution metrics, KPIs, and compliance dashboards for bottom-up reporting. This ensures follow-up campaigns remain traceable to their originating commitment and are accurately represented in effectiveness reporting.
Does Quartica MARS support aRMM material authoring and country-specific translation workflows?
Yes. Quartica MARS supports structured authoring of aRMM materials and can generate country-specific variants aligned to Core and Local Implementation Plans using AI-assisted authoring capabilities. Translation workflows can be integrated into the material lifecycle, preserving version linkage between global source content and local language adaptations throughout the governance chain.
Does Quartica MARS replace existing RIM, safety, or medical review systems?
No. Quartica MARS integrates with regulatory, safety, quality, and medical review systems to extend governance and traceability across aRMM activities. It does not replace established systems of record; it connects them within a structured risk commitment framework through secure, API-driven bidirectional data exchange.
Can Quartica MARS support ad-hoc safety communications such as DHPCs alongside planned aRMM activities?
Yes. Quartica MARS supports urgent and ad-hoc communications including Direct Healthcare Professional Communications (DHPCs) within the same governed framework as planned aRMM activities covering drafting, approval workflows, distribution, and acknowledgement tracking. All risk-related communications, planned or unplanned, are managed within a single validated platform with consistent traceability and audit-ready records.
Can Quartica MARS scale across multiple products, regions, and alliance partner models?
Yes. The architecture supports multi-product, multi-region, and alliance partner models within a single validated framework. Organizations with complex global pipelines can manage Core and Local commitments, materials, and distribution records across markets without separate instances or manual consolidation with QPPV oversight dashboards reflecting completeness and compliance status across the full portfolio.

